|
11/6/2023 0 Comments Na element or compoundQuestion 1: What is the nature of Sodium Hydroxide? 
Can be used for the extraction of zinc and plating of tin.Sodium hydroxide plays an important role in the Bayer process which is used to produce aluminium.Used to refine petroleum in the petroleum industry.It can be used to clean drains in their concentrated form.Mainly used in the soap manufacturing process.The melting point of sodium hydroxide is 318.4 ο Celsius.Sodium hydroxide is insoluble in ether.Can be easily dissolved in water or alcohol.It has a high boiling point of 1390 ο Celsius.Now sodium amalgam is treated with water to get sodium hydroxide or caustic soda along with hydrogen gas and mercury. The process can be easily understood by the following reactions at anode and cathode. Formation of sodium amalgam takes place at cathode and chlorine gas is released at anode. 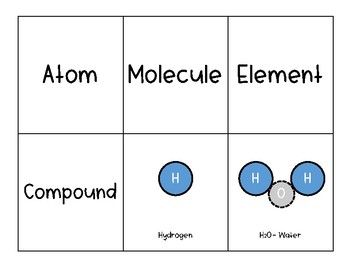
Take mercury as cathode and carbon rode as an anode, sodium chloride solution is now electrolyzed. It is prepared by Sodium chloride electrolysis which is performed in a special type of cell called the Castner-Kellner cell. Sodium Hydroxide is also known as Caustic Soda and its formula is NaOH.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
