|
One mole of electric charge (96,500 coulombs), when passed through a cell, will discharge half a mole of a divalent metal ion such as Cu 2 +. Most hydrogen gas is manufactured by the steam reforming of natural gas. However, it becomes more efficient at higher temperatures, where thermal energy reduces the amount of electrical energy required, so there is now some interest in developing high-temperature electrolytic processes. This process is generally too expensive for industrial production unless highly pure hydrogen is required. Electrolysis of a solution of sulfuric acid or of a salt such as NaNO 3 results in the decomposition of water at both electrodes:Įlectrolytic production of hydrogen is usually carried out with a dilute solution of sulfuric acid. If the object is to produce hydrogen and oxygen, the electrolyte must be energetically more difficult to oxidize or reduce than water itself. Pure water is an insulator and cannot undergo significant electrolysis without adding an electrolyte. Only those ions that are near the interfacial region are likely to undergo migration. Migration- the motion of a charged particle due to an applied electric field, is only a minor player, producing only about one non-random jump out of around 100,000 random ones for a 1 volt cm –1 electric field. Ionic motion throughout the bulk of the solution occurs mostly by diffusion, which is the transport of molecules in response to a concentration gradient. 
This is the only fundamental difference between an electrolytic cell and the galvanic cell in which the free energy supplied by the cell reaction is extracted as work done on the surroundings.Ī common misconception about electrolysis is that "ions are attracted to the oppositely-charged electrode." This is true only in the very thin interfacial region near the electrode surface. The free energy is supplied in the form of electrical work done on the system by the outside world (the surroundings). Thus if a solution of nickel chloride undergoes electrolysis at platinum electrodes, the reactions are Ni 2 + + 2 e – → Ni(s)īoth of these processes are carried out in electrochemical cells which are forced to operate in the "reverse", or non-spontaneous direction, as indicated by the negative for the above cell reaction. Ions in aqueous solutions can undergo similar reactions. The electrolysis of sodium and potassium hydroxides, first carried out in 1808 by Sir Humphrey Davey, led to the discovery of these two metallic elements and showed that these two hydroxides which had previously been considered un-decomposable and thus elements, were in fact compounds.Įlectrolysis of molten alkali halides is the usual industrial method of preparing the alkali metals: Na + + e – → Na (l) The largest of these are the chloralkali industry (chlorine and "caustic"), and the refining of aluminum the latter consumes approximately 5% of the electrical power generated in North America.Įlectrolysis refers to the decomposition of a substance by an electric current. A large number of electrolysis processes are employed by industry to refine metals and to produce both inorganic and organic products.Thus an attempt to electrolyze a solution of NaNO 3 will produce only H 2 and O 2. When an aqueous solution is subjected to electrolysis, the oxidation or reduction of water can be a competing process and may dominate if the applied voltage is sufficiently great.Ionic transport in the greater part of the electrolyte is by ordinary thermal diffusion- the statistical tendency of concentrations to become uniform. Transport of ions in the electrolyte in response to the potential difference between the electrodes ("drift") is largely restricted to the regions very close to the electrodes.In practice, the products of electrolysis are usually simpler than the reactants, hence the term electro- lysis. An electrolytic cell is in this sense the opposite of a galvanic cell. 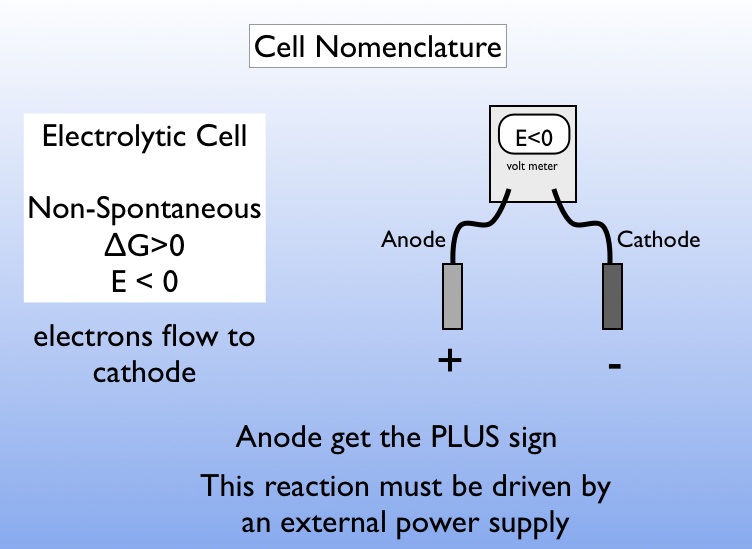
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
